Absolute Bioavailability
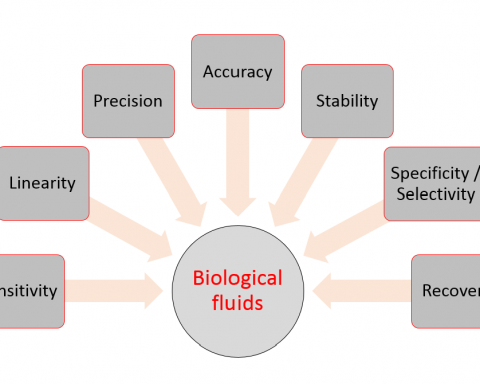
The absolute bioavailability of a given drug from a dosage form is the fraction (or percentage) of the administered dose which is absorbed intact into the systemic circulation. Absolute bioavailability may be calculated by comparing the total amount of intact drug that reaches the systemic circulation after the administration of a known dose of the dosage form via a route of administration, with the total amount that reaches the systemic circulation after the administration of an equivalent dose of the drug in the form of an intravenous bolus injection. An intravenous bolus injection is used as a reference to compare the systemic availability of the drug administered via different routes, because when a drug is delivered intravenously the entire administered dose is introduced directly into the systemic circulation, i.e. it has no absorption barriers to cross and is therefore considered to be totally bioavailable.
Relative Bioavailability
In the case of drugs that cannot be administered by intravenous bolus injection, the relative (or comparative) bioavailability is determined rather than the absolute bioavailability. In this case the bioavailability of a given drug from a ‘test’ dosage form is compared to that of the same drug administered in a ‘standard’ dosage form, which is either an orally administered solution (from which the drug is known to be well absorbed) or an established commercial preparation of proven clinical effectiveness. Hence relative bioavailability is a measure of the fraction (or percentage) of a given drug that is absorbed intact into the systemic circulation from a dosage form relative to a recognized (i.e. clinically proven) standard dosage form of that drug.